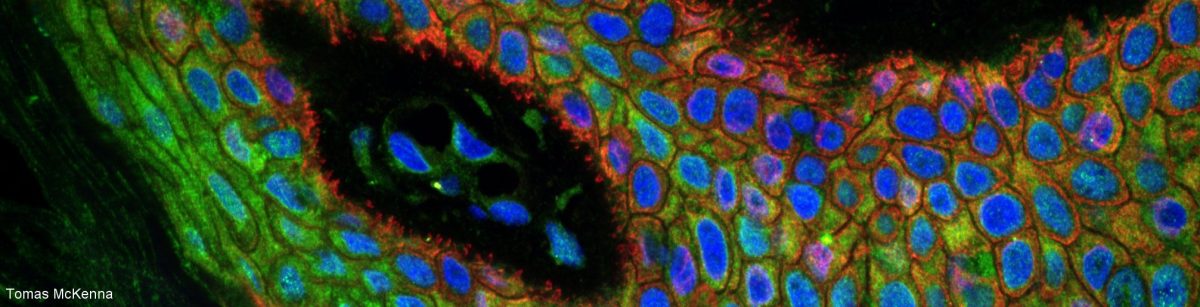
9th of September, 10-12am: Live/zoom demo of the latest microscope acquired by the LCI facility: Crest spinning disk with DeepSIM (new Orion)
- Ti2 microscope
- Crest spinning disk (identical to the one on Tweety but with slightly different wavelengths)
- DeepSIM: SIM is a fast super-resolution technique which is gentle for the sample (less bleaching, less photodamage for live samples) and offers double the resolution of a confocal microscope (down to 120nm resolution), even thick in samples. It is flexible and can be used with any objectives, even low magnification air objectives.
- No specific sample preparation requirements
- More information here
For the best experience, join the demo in person at the LCI facility (please apply by replying to this email) but if you cannot make it, you can also listen on Zoom.
The system has been purchased by the LCI so it is here to stay! 🙂
Please let us know if you want to join the demo in person and mention if you would like to try imaging your own sample with DeepSIM.